| << Chapter < Page | Chapter >> Page > |
An acid-base reaction is one in which a hydrogen ion, H + , is transferred from one chemical species to another. Such reactions are of central importance to numerous natural and technological processes, ranging from the chemical transformations that take place within cells and the lakes and oceans, to the industrial-scale production of fertilizers, pharmaceuticals, and other substances essential to society. The subject of acid-base chemistry, therefore, is worthy of thorough discussion, and a full chapter is devoted to this topic later in the text.
For purposes of this brief introduction, we will consider only the more common types of acid-base reactions that take place in aqueous solutions. In this context, an acid is a substance that will dissolve in water to yield hydronium ions, H 3 O + . As an example, consider the equation shown here:
The process represented by this equation confirms that hydrogen chloride is an acid. When dissolved in water, H 3 O + ions are produced by a chemical reaction in which H + ions are transferred from HCl molecules to H 2 O molecules ( [link] ).
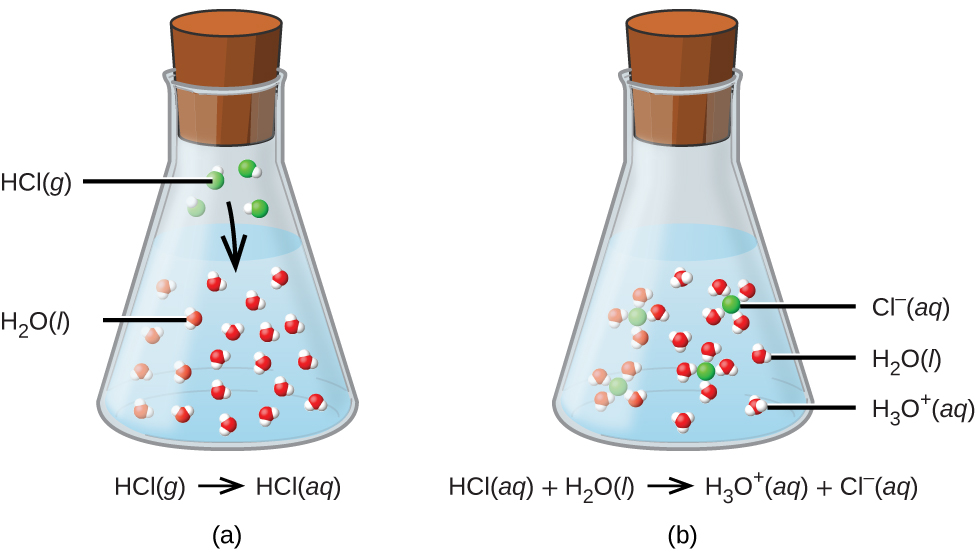
The nature of HCl is such that its reaction with water as just described is essentially 100% efficient: Virtually every HCl molecule that dissolves in water will undergo this reaction. Acids that completely react in this fashion are called strong acids , and HCl is one among just a handful of common acid compounds that are classified as strong ( [link] ). A far greater number of compounds behave as weak acids and only partially react with water, leaving a large majority of dissolved molecules in their original form and generating a relatively small amount of hydronium ions. Weak acids are commonly encountered in nature, being the substances partly responsible for the tangy taste of citrus fruits, the stinging sensation of insect bites, and the unpleasant smells associated with body odor. A familiar example of a weak acid is acetic acid, the main ingredient in food vinegars:
When dissolved in water under typical conditions, only about 1% of acetic acid molecules are present in the ionized form, ( [link] ). (The use of a double-arrow in the equation above denotes the partial reaction aspect of this process, a concept addressed fully in the chapters on chemical equilibrium.)
| Common Strong Acids | |
|---|---|
| Compound Formula | Name in Aqueous Solution |
| HBr | hydrobromic acid |
| HCl | hydrochloric acid |
| HI | hydroiodic acid |
| HNO 3 | nitric acid |
| HClO 4 | perchloric acid |
| H 2 SO 4 | sulfuric acid |
A base is a substance that will dissolve in water to yield hydroxide ions, OH − . The most common bases are ionic compounds composed of alkali or alkaline earth metal cations (groups 1 and 2) combined with the hydroxide ion—for example, NaOH and Ca(OH) 2 . When these compounds dissolve in water, hydroxide ions are released directly into the solution. For example, KOH and Ba(OH) 2 dissolve in water and dissociate completely to produce cations (K + and Ba 2+ , respectively) and hydroxide ions, OH − . These bases, along with other hydroxides that completely dissociate in water, are considered strong bases .

Notification Switch
Would you like to follow the 'Chemistry' conversation and receive update notifications?