Comprehensive art program
Our art program is designed to enhance students’ understanding of concepts through clear, effective illustrations, diagrams, and photographs.
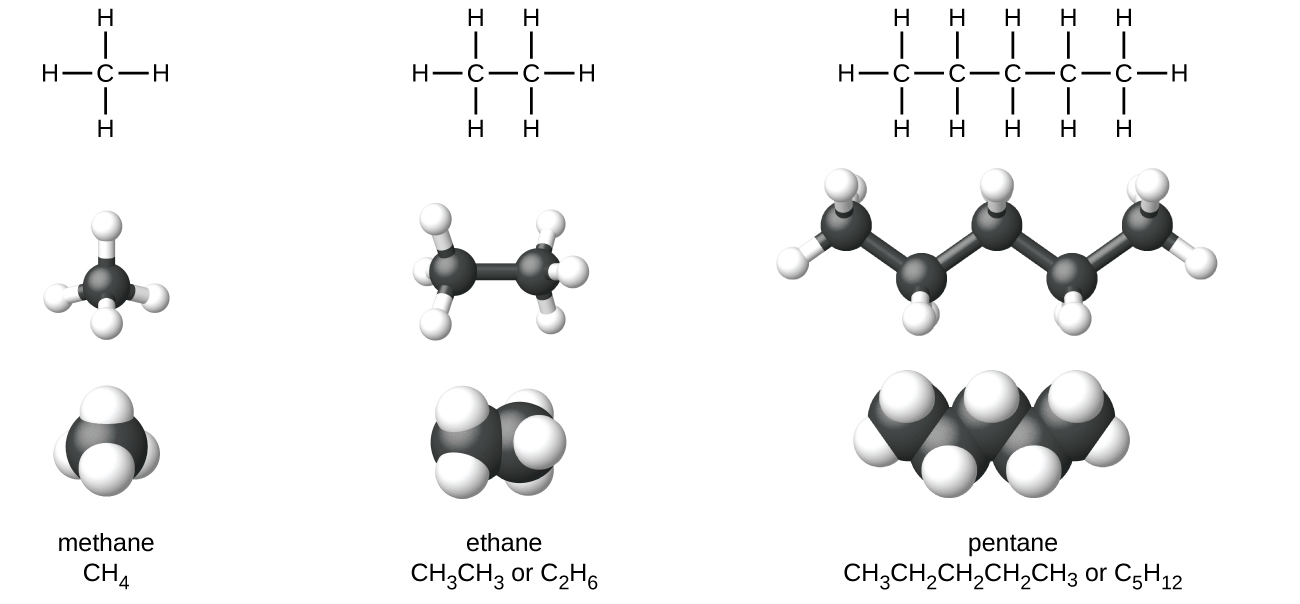
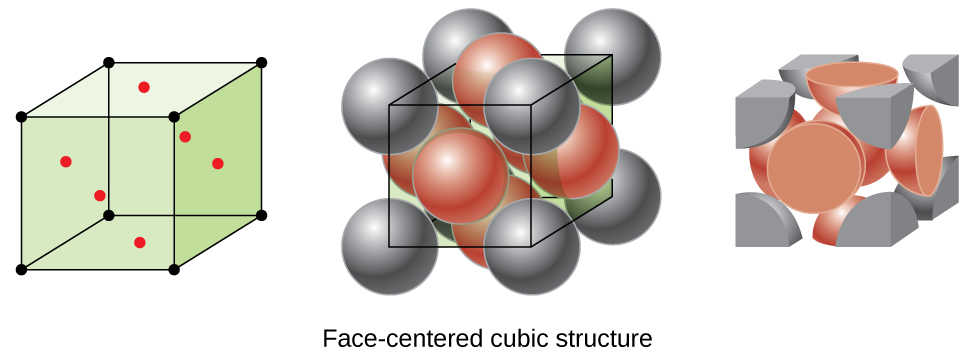
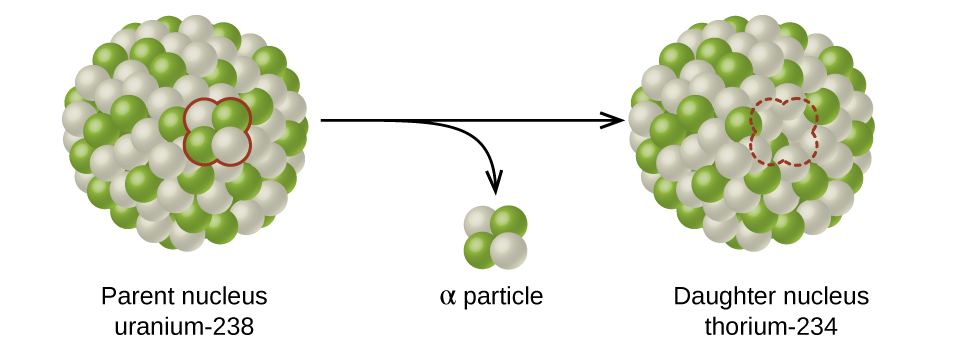
Interactives that engage
Chemistry incorporates links to relevant interactive exercises and animations that help bring topics to life through our
Link to Learning feature. Examples include:
- PhET simulations
- IUPAC data and interactives
- TED talks
Assessments that reinforce key concepts
In-chapter
Examples walk students through problems by posing a question, stepping out a solution, and then asking students to practice the skill with a “Check Your Learning” component. The book also includes assessments at the end of each chapter so students can apply what they’ve learned through practice problems.
Atom-first alternate sequencing
Chemistry was conceived and written to fit a particular topical sequence, but it can be used flexibly to accommodate other course structures. Some instructors prefer to organize their course in a molecule-first or atom-first organization. For professors who use this approach, our OpenStax
Chemistry textbook can be sequenced to fit this pedagogy. Please consider, however, that the chapters were not written to be completely independent, and that the proposed alternate sequence should be carefully considered for student preparation and textual consistency. We recommend these shifts in the table of contents structure if you plan to create a molecule/atom-first version of this text for your students:
- Chapter 1: Essential Ideas
- Chapter 2: Atoms, Molecules, and Ions
- Chapter 6: Electronic Structure and Periodic Properties of Elements
- Chapter 7: Chemical Bonding and Molecular Geometry
- Chapter 8: Advanced Theories of Covalent Bonding
- Chapter 3: Composition of Substances and Solutions
- Chapter 4: Stoichiometry of Chemical Reactions
- Chapter 5: Thermochemistry
- Chapter 9: Gases
- Chapter 10: Liquids and Solids
- Chapter 11: Solutions and Colloids
- Chapter 12: Kinetics
- Chapter 13: Fundamental Equilibrium Concepts
- Chapter 14: Acid-Base Equilibria
- Chapter 15: Equilibria of Other Reaction Classes
- Chapter 16: Thermodynamics
- Chapter 17: Electrochemistry
- Chapter 18: Representative Metals, Metalloids, and Nonmetals
- Chapter 19: Transition Metals and Coordination Chemistry
- Chapter 20: Organic Chemistry
- Chapter 21: Nuclear Chemistry
Ancillaries
OpenStax projects offer an array of ancillaries for students and instructors. The following resources are available.
-
PowerPoint Slides
-
Instructor’s Solution Manual
Our resources are continually expanding, so please visit http://openstaxcollege.org to view an up-to-date list of the Learning Resources for this title and to find information on accessing these resources.
About our team
Content leads
Paul Flowers, PhD, University of North Carolina - Pembroke
Dr. Paul Flowers earned a BS in Chemistry from St. Andrews Presbyterian College in 1983 and a PhD in Analytical Chemistry from the University of Tennessee in 1988. After a one-year postdoctoral appointment at Los Alamos National Laboratory, he joined the University of North Carolina–Pembroke in the fall of 1989. Dr. Flowers teaches courses in general and analytical chemistry, and conducts experimental research involving the development of new devices and methods for microscale chemical analysis.
Klaus Theopold, PhD, University of Delaware
Dr. Klaus Theopold (born in Berlin, Germany) received his Vordiplom from the Universität Hamburg in 1977. He then decided to pursue his graduate studies in the United States, where he received his PhD in inorganic chemistry from UC Berkeley in 1982. After a year of postdoctoral research at MIT, he joined the faculty at Cornell University. In 1990, he moved to the University of Delaware, where he is a Professor in the Department of Chemistry and Biochemistry and serves as an Associate Director of the University’s Center for Catalytic Science and Technology. Dr. Theopold regularly teaches graduate courses in inorganic and organometallic chemistry as well as General Chemistry.
Richard Langley, PhD, Stephen F. Austin State University
Dr. Richard Langley earned BS degrees in Chemistry and Mineralogy from Miami University of Ohio in the early 1970s and went on to receive his PhD in Chemistry from the University of Nebraska in 1977. After a postdoctoral fellowship at the Arizona State University Center for Solid State Studies, Dr. Langley taught in the University of Wisconsin system and participated in research at Argonne National Laboratory. Moving to Stephen F. Austin State University in 1982, Dr. Langley today serves as Professor of Chemistry. His areas of specialization are solid state chemistry, synthetic inorganic chemistry, fluorine chemistry, and chemical education.
Senior contributing author
William R. Robinson, PhD
Contributing authors
Mark Blaser, Shasta College
Simon Bott, University of Houston
Donald Carpenetti, Craven Community College
Andrew Eklund, Alfred University
Emad El-Giar, University of Louisiana at Monroe
Don Frantz, Wilfrid Laurier University
Paul Hooker, Westminster College
Jennifer Look, Mercer University
George Kaminski, Worcester Polytechnic Institute
Carol Martinez, Central New Mexico Community College
Troy Milliken, Jackson State University
Vicki Moravec, Trine University
Jason Powell, Ferrum College
Thomas Sorensen, University of Wisconsin–Milwaukee
Allison Soult, University of Kentucky
Contributing reviewers
Casey Akin, College Station Independent School District
Lara AL-Hariri, University of Massachusetts–Amherst
Sahar Atwa, University of Louisiana at Monroe
Todd Austell, University of North Carolina–Chapel Hill
Bobby Bailey, University of Maryland–University College
Robert Baker, Trinity College
Jeffrey Bartz, Kalamazoo College
Greg Baxley, Cuesta College
Ashley Beasley Green, National Institute of Standards and Technology
Patricia Bianconi, University of Massachusetts
Lisa Blank, Lyme Central School District
Daniel Branan, Colorado Community College System
Dorian Canelas, Duke University
Emmanuel Chang, York College
Carolyn Collins, College of Southern Nevada
Colleen Craig, University of Washington
Yasmine Daniels, Montgomery College–Germantown
Patricia Dockham, Grand Rapids Community College
Erick Fuoco, Richard J. Daley College
Andrea Geyer, University of Saint Francis
Daniel Goebbert, University of Alabama
John Goodwin, Coastal Carolina University
Stephanie Gould, Austin College
Patrick Holt, Bellarmine University
Kevin Kolack, Queensborough Community College
Amy Kovach, Roberts Wesleyan College
Judit Kovacs Beagle, University of Dayton
Krzysztof Kuczera, University of Kansas
Marcus Lay, University of Georgia
Pamela Lord, University of Saint Francis
Oleg Maksimov, Excelsior College
John Matson, Virginia Tech
Katrina Miranda, University of Arizona
Douglas Mulford, Emory University
Mark Ott, Jackson College
Adrienne Oxley, Columbia College
Richard Pennington, Georgia Gwinnett College
Rodney Powell, Coastal Carolina Community College
Jeanita Pritchett, Montgomery College–Rockville
Aheda Saber, University of Illinois at Chicago
Raymond Sadeghi, University of Texas at San Antonio
Nirmala Shankar, Rutgers University
Jonathan Smith, Temple University
Bryan Spiegelberg, Rider University
Ron Sternfels, Roane State Community College
Cynthia Strong, Cornell College
Kris Varazo, Francis Marion University
Victor Vilchiz, Virginia State University
Alex Waterson, Vanderbilt University
JuchaoYan, Eastern New Mexico University
Mustafa Yatin, Salem State University
Kazushige Yokoyama, State University of New York at Geneseo
Curtis Zaleski, Shippensburg University
Wei Zhang, University of Colorado–Boulder







