-
Home
- Chemistry
- Equilibria of other reaction
- Lewis acids and bases
Dissociation of a complex ion
Calculate the concentration of the silver ion in a solution that initially is 0.10
M with respect to
Solution
We use the familiar path to solve this problem:
-
Determine the direction of change. The complex ion
is in equilibrium with its components, as represented by the equation:
We write the equilibrium as a formation reaction because
Appendix K lists formation constants for complex ions. Before equilibrium, the reaction quotient is larger than the equilibrium constant [
K
f = 1.7
10
7 , and
it is infinitely large], so the reaction shifts to the left to reach equilibrium.
-
Determine x
and equilibrium concentrations. We let the change in concentration of Ag
+ be
x . Dissociation of 1 mol of
gives 1 mol of Ag
+ and 2 mol of NH
3 , so the change in [NH
3 ] is 2
x and that of
is –
x . In summary:
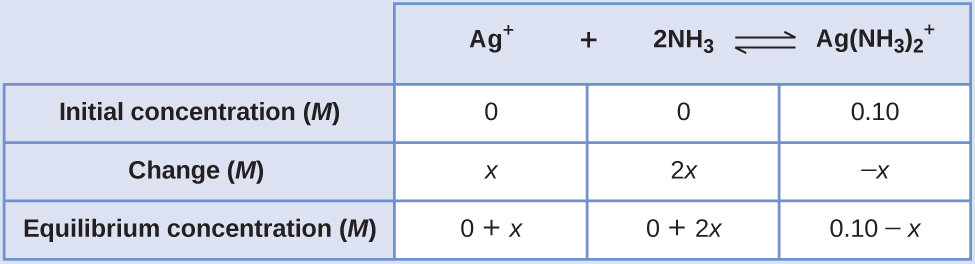
-
Solve for x and the equilibrium concentrations. At equilibrium:
Both
Q and
K
f are much larger than 1, so let us assume that the changes in concentrations needed to reach equilibrium are small. Thus 0.10 –
x is approximated as 0.10:
Because only 1.1% of the
dissociates into Ag
+ and NH
3 , the assumption that
x is small is justified.
Now we determine the equilibrium concentrations:
The concentration of free silver ion in the solution is 0.0011
M .
-
Check the work. The value of
Q calculated using the equilibrium concentrations is equal to
K
f within the error associated with the significant figures in the calculation.
Check your learning
Calculate the silver ion concentration, [Ag
+ ], of a solution prepared by dissolving 1.00 g of AgNO
3 and 10.0 g of KCN in sufficient water to make 1.00 L of solution. (Hint: Because
Q <
K
f , assume the reaction goes to completion then calculate the [Ag
+ ] produced by dissociation of the complex.)
Got questions? Get instant answers now!
Key concepts and summary
G.N. Lewis proposed a definition for acids and bases that relies on an atom’s or molecule’s ability to accept or donate electron pairs. A Lewis acid is a species that can accept an electron pair, whereas a Lewis base has an electron pair available for donation to a Lewis acid. Complex ions are examples of Lewis acid-base adducts. In a complex ion, we have a central atom, often consisting of a transition metal cation, which acts as a Lewis acid, and several neutral molecules or ions surrounding them called ligands that act as Lewis bases. Complex ions form by sharing electron pairs to form coordinate covalent bonds. The equilibrium reaction that occurs when forming a complex ion has an equilibrium constant associated with it called a formation constant,
K
f . This is often referred to as a stability constant, as it represents the stability of the complex ion. Formation of complex ions in solution can have a profound effect on the solubility of a transition metal compound.
Questions & Answers
evolutionary history and relationship of an organism or group of organisms
AI-Robot
the study of living organisms and their interactions with one another and their environments
AI-Robot
HOW CAN MAN ORGAN FUNCTION
the diagram of the digestive system
allimentary cannel
Ogenrwot
They formed in two ways first when one sperm and one egg are splited by mitosis or two sperm and two eggs join together
Oluwatobi
Genetics is the study of heredity
Misack
how does twins formed?
Misack
discuss biological phenomenon and provide pieces of evidence to show that it was responsible for the formation of eukaryotic organelles
the study of living organisms and their interactions with one another and their environment.
Wine
discuss the biological phenomenon and provide pieces of evidence to show that it was responsible for the formation of eukaryotic organelles in an essay form
list any five characteristics of the blood cells
Shaker
lack electricity and its more savely than electronic microscope because its naturally by using of light
advantage of electronic microscope is easily and clearly while disadvantage is dangerous because its electronic. advantage of light microscope is savely and naturally by sun while disadvantage is not easily,means its not sharp and not clear
Abdullahi
cell theory state that every organisms composed of one or more cell,cell is the basic unit of life
Abdullahi
is like gone fail us
DENG
cells is the basic structure and functions of all living things
Ramadan
is organisms that are similar into groups called tara
Yamosa
in what situation (s) would be the use of a scanning electron microscope be ideal and why?
A scanning electron microscope (SEM) is ideal for situations requiring high-resolution imaging of surfaces. It is commonly used in materials science, biology, and geology to examine the topography and composition of samples at a nanoscale level. SEM is particularly useful for studying fine details,
Hilary
Got questions? Join the online conversation and get instant answers!
Source:
OpenStax, Chemistry. OpenStax CNX. May 20, 2015 Download for free at http://legacy.cnx.org/content/col11760/1.9
Google Play and the Google Play logo are trademarks of Google Inc.


