| << Chapter < Page | Chapter >> Page > |
Watch this video explaining how a muscle contraction is signaled.
Which of the following statements about muscle contraction is true?
View this animation of the cross-bridge muscle contraction.
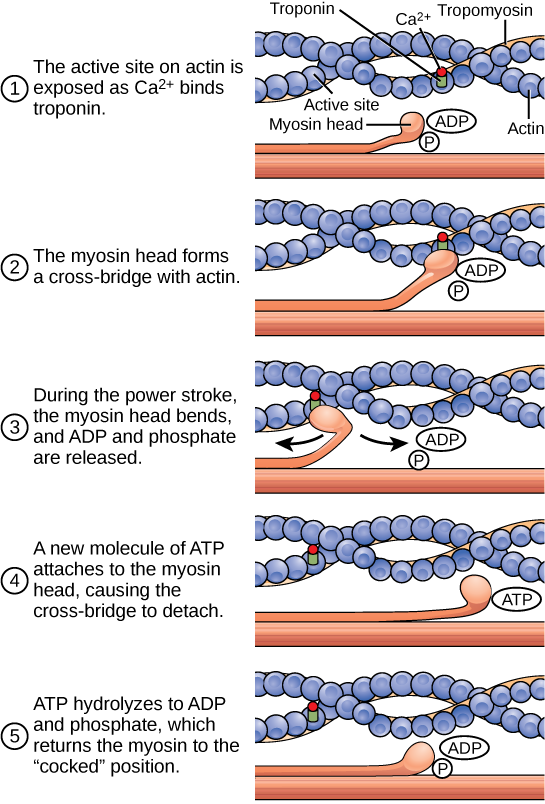
When a muscle is in a resting state, actin and myosin are separated. To keep actin from binding to the active site on myosin, regulatory proteins block the molecular binding sites. Tropomyosin blocks myosin binding sites on actin molecules, preventing cross-bridge formation and preventing contraction in a muscle without nervous input. Troponin binds to tropomyosin and helps to position it on the actin molecule; it also binds calcium ions.
To enable a muscle contraction, tropomyosin must change conformation, uncovering the myosin-binding site on an actin molecule and allowing cross-bridge formation. This can only happen in the presence of calcium, which is kept at extremely low concentrations in the sarcoplasm. If present, calcium ions bind to troponin, causing conformational changes in troponin that allow tropomyosin to move away from the myosin binding sites on actin. Once the tropomyosin is removed, a cross-bridge can form between actin and myosin, triggering contraction. Cross-bridge cycling continues until Ca 2+ ions and ATP are no longer available and tropomyosin again covers the binding sites on actin.
Excitation–contraction coupling is the link (transduction) between the action potential generated in the sarcolemma and the start of a muscle contraction. The trigger for calcium release from the sarcoplasmic reticulum into the sarcoplasm is a neural signal. Each skeletal muscle fiber is controlled by a motor neuron, which conducts signals from the brain or spinal cord to the muscle. The area of the sarcolemma on the muscle fiber that interacts with the neuron is called the motor end plate . The end of the neuron’s axon is called the synaptic terminal, and it does not actually contact the motor end plate. A small space called the synaptic cleft separates the synaptic terminal from the motor end plate. Electrical signals travel along the neuron’s axon, which branches through the muscle and connects to individual muscle fibers at a neuromuscular junction.
The ability of cells to communicate electrically requires that the cells expend energy to create an electrical gradient across their cell membranes. This charge gradient is carried by ions, which are differentially distributed across the membrane. Each ion exerts an electrical influence and a concentration influence. Just as milk will eventually mix with coffee without the need to stir, ions also distribute themselves evenly, if they are permitted to do so. In this case, they are not permitted to return to an evenly mixed state.

Notification Switch
Would you like to follow the 'Biology' conversation and receive update notifications?